IZiel works in collaboration with your team to develop the complete Design History File (DHF) including requirements management, risk management, process validations and software validations using robust design controls process and quality system procedures. Thereafter, IZiel team works with their regulatory team in USA to complete the submissions (510k or PMA) and resolve queries, if any, from USFDA.
USFDA Approvals:
Upon completion of Engineering & QMS Documentation, the regulatory team of IZiel conducts the predicate device search, comparative analysis and ensure all the necessary standards are complied with. Thereafter, IZiel team initiate the writing of 510 (k) / PMA, which is verified and approved by our US Regulatory Consultants. Our highly experienced US regulatory consultants include Ex-FDA Auditors, have conducted 3500 + reviews and approvals and have experience with Class I, II & III devices in Cardiology, Neurology, Image Diagnostics, Surgical, In-Vitro Diagnostics, Wound Dressing etc
Remediation:
Medical Device Manufacturers receive observations, warnings &/or 483s that require substantial number of unplanned resources, budgets & at the worst are declined an opportunity to sell until the observations are cleared. Remediation is a set of activities or a project to resolve these observations & warnings. Companies may also undertake product remediation to resolve issues that they are aware of in their product design and development process.
Remediation requires the application of best practices in design and development while ensuring compliance with regulation to ensure a speedy and compliant design and development process.
Typical approach and the topics covered by the IZiel team in collaboration with the customer is depicted as below –
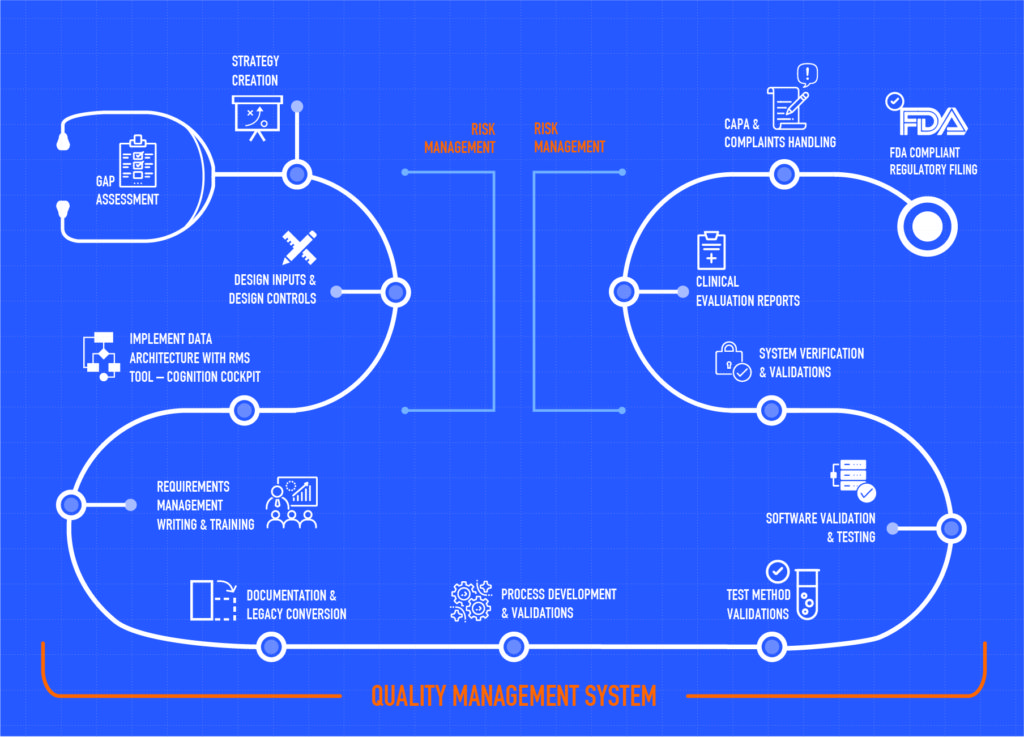
IZiel can help companies understand the gaps in their design and development process in a methodical and comprehensive manner. Once the gaps are identified, we work with companies in a flexible on-shore/off-shore model to close these gaps, while ensuring costs are kept manageable.
IZiel can provide quick scalability of the skilled resources with knowledge of Design, Process Engineering & Validation, Systems Engineering, QMS and Regulatory to achieve outcome-based deliveries. The IZiel team comprises of experts in the Medical Device Industry and has a highly trained and experienced off-shore team. This team not only consults but also helps in implementation of the solutions identified.
Our founding principles to work for remediation projects –