Acquisition Integration of Medical Devices
Mergers & Acquisition in the healthcare industry has been strong in recent years and is expected to grow. Acquiring a new company or product line can have a positive business impact, but it also comes with implementation, process, quality & regulatory challenges. Especially, in the medical device industry, failure to comply with regulations can result in the loss of authority to deliver products and services.
IZiel enables the company to ensure compliance with the product specific standards & requirements by performing high-quality due diligence to mitigate risk and thereafter provide support to complete documentation as per USFDA / EUMDR requirements. IZiel has successfully worked with one of the world’s largest medical device manufacturers to update more than 2000 documents, 500+ approvals with release in Agile in a short span of 8-9 months.
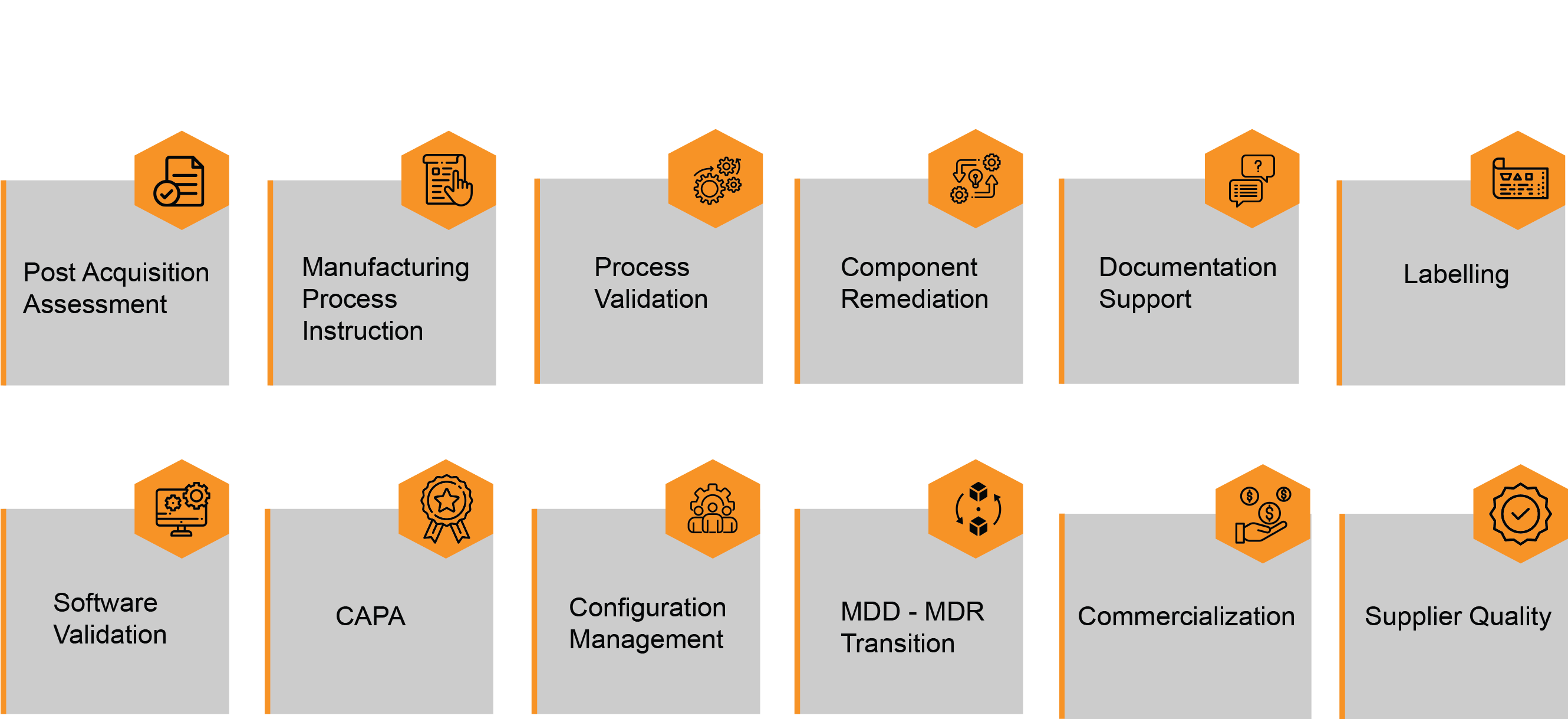
IZiel Approach – Acquisition Integration of Medical Devices
Strong Project Management, Medical device Expertise, Quick Scalability of Skilled Resources are the key enablers of our success for Acquisition Integration Projects. As depicted above, IZiel’s approach has been very comprehensive and methodical. IZiel team can support your team in various ways, including but not restricted to –
Post-Acquisition Assessment – IZiel Team works with your acquisition integration team to conduct a detailed Gap Assessment to define acquisition high risk areas from a compliance standpoint. Assessment include QMS Assessment, risk classification for key components/processes, task prioritization, planning & integration support to upgrade the documentation to parent company’s documentation and quality standards.
Manufacturing Process Instructions – Updated wordy and incoherent MPI’s into the company format by incorporating images, clarifying requirements etc. Ensure complete coherence in Practice vs Procedure (PvP) by creating redlines and change justification to ensure PvP Gap closure.
Process Optimization & Validation – IZiel Experts are DFSS Master Black Belts that can help your team to reduce the cycle time, wastage etc. IZiel Team works with the customer to complete FAT, SAT, IQ, OQ, PQ, TMD, & TMV alongwith other process validation activities.
Labelling – IZiel Team has developed an integrated Labelling Model to update/develop large number of EUMDR compliant labels in a fast and effective manner. We assist in reviewing the label samples and specification for accuracy of content including but not limited to product descriptions, regulatory symbols, translations, size specifications, UDI data, branding information. IZiel has experience with Bartender Label Design Software, Barcode Verification Equipment’s etc. to ensure the labels meet the requirements of internal QS and external standards.
Software & Tool Validation – IZiel Team would perform complete Software Verification and Validation as per IEC 62304 & IEC 82304 including requirements management, architecture design, configuration management, risk mitigations, unit & integration testing, code based static analysis, verification traceability, software release and software maintenance activities.
IZiel utilizes the GAMP 5 Categorization to complete Tool Validation (CSV) for various non-product & enterprise level software.
Documentation Support – IZiel has successfully delivered various projects through the
Onshore-Offshore Work Model thereby completing projects faster and with major cost savings. IZiel team works hand-in-hand with the client to plan, execute, compile reports and receive stakeholder approvals for documents completed. IZiel has successfully worked with one of the world’s largest medical device manufacturers to update more than 2000 documents, 500+ approvals with release in Agile in a short span of 8-9 months.
Configuration Management – IZiel employs a group of document specialists that have expertise in QMS architecture for both product lifecycle processes and enabling processes, including but not limited to change control, document management, process deviations, and supplier change management.
Component Remediation – We at IZiel support and manage all component remediation activities including but not limited to identification of CTQ (critical-to-quality) features, quality impact assessments, component qualifications, FLQIA, PLQIA, Spec Updates and component Test Method Development (TMD) and Validation (TMV).
CAPA – IZiel covers all the product-specific requirements for all components, manufacturing processes, verification & validation along with corrective and preventative actions (CAPA) for assessing customer satisfaction, product non-conformance, assessing and improving quality policies and procedures, carrying out and assessing the results of internal audits, and implementing systems for continuous improvement. IZiel Team has successfully demonstrated capabilities as CAPA Owners for various CAPAs on Process Validation, PVP, Internal Audit NC, Operator Certification and Software Validation.
Commercialization – We at IZiel support and manage all Commercialization activities including creation of GTINs, BOM’s, DHR’s, Model Numbers, Labels, and all other supporting documentation as well as GTIN release activities such as product release authorizations and market authorizations.
Supplier Quality – IZiel supports and manages supplier quality activities including supplier selection and evaluation, ASL (approved supplier list) management, supplier change management, supplier segmentation and classification.
Why to Choose IZiel?
At IZiel, our team has upgraded thousands of documents for various acquisition integration projects of Class I, II & III medical device companies. Our Outcome Based & Onshore-Offshore Delivery Model has worked effectively to complete the Post-Merger Integration with significant reduction in timelines and budgets.